This is high enough to permit a sustained nuclear fission chain reaction to occur, provided other supporting geologic conditions exist. The ore deposit is 1.7 billion years old at that time, uranium-235 constituted about 3% of the total uranium on Earth (0.72% today). In 1972 French physicist Francis Perrin discovered ancient and no longer active prehistoric natural nuclear fission reactors in uranium ore deposits at the Oklo mine in Gabon, West Africa, collectively known as the Oklo Fossil Reactors. The security of those weapons and their fissile material following the breakup of the Soviet Union in 1991 is an ongoing concern. In 1934 research by Enrico Fermi and others eventually led to the use of uranium fission in the first nuclear weapon used in war and later in the peaceful use of uranium as fuel in nuclear power production. An ensuing arms race during the Cold War between the United States and the Soviet Union produced tens of thousands of nuclear weapons that used uranium metal and uranium-derived plutonium-239. Becquerel discovered that uranium exhibited invisible light or rays it was radioactivity. The pure metal was first isolated in 1841 by Eugène-Melchior Péligot, who reduced anhydrous uranium tetrachloride with potassium metal. What Klaproth actually identified was not the pure element but uranium oxide. While experimenting with pitchblende in 1789, he concluded that it contained a new element, which he named after the newly discovered planet Uranus (named after the Greek god of the sky or heaven). The identification of uranium as an element is generally credited to Martin H. In the late Middle Ages, pitchblende was extracted from the silver mines and was used as a coloring agent in the glassmaking industry. Yellow glass with 1% uranium oxide was found in an ancient Roman villa near Naples, Italy. when it was used as a yellow coloring agent in ceramic glazes. The use of uranium in its natural oxide form dates back to 79 A.D. Uranium-235 has a half-life of 700 million years.Uranium(VI) oxides or "yellow cake" is an intermediate step in the processing of uranium ores. Most modern nuclear arsenals use plutonium as the fissile component, however U-235 devices remain a nuclear proliferation concern due to the simplicity of the design. The Little Boy atomic bomb was fueled by enriched uranium. However, judicious use of implosion and neutron reflectors can enable construction of a weapon from a quantity of uranium below the usual critical mass for its level of enrichment, though this would likely only be possible in a country which already had extensive experience in developing nuclear weapons. The fissile uranium in nuclear weapons usually contains 85% or more of 235U known as weapon(s)-grade, though for a crude, inefficient weapon 20% is sufficient (called weapon(s)-usable) even less is sufficient, but then the critical mass required rapidly increases. Uranium which has been processed to boost its uranium-235 proportion is known as enriched uranium, different applications require unique levels of enrichment. 
Pressurised Heavy Water Reactors, other heavy water reactors, and some graphite moderated reactors are known for using unenriched uranium. 
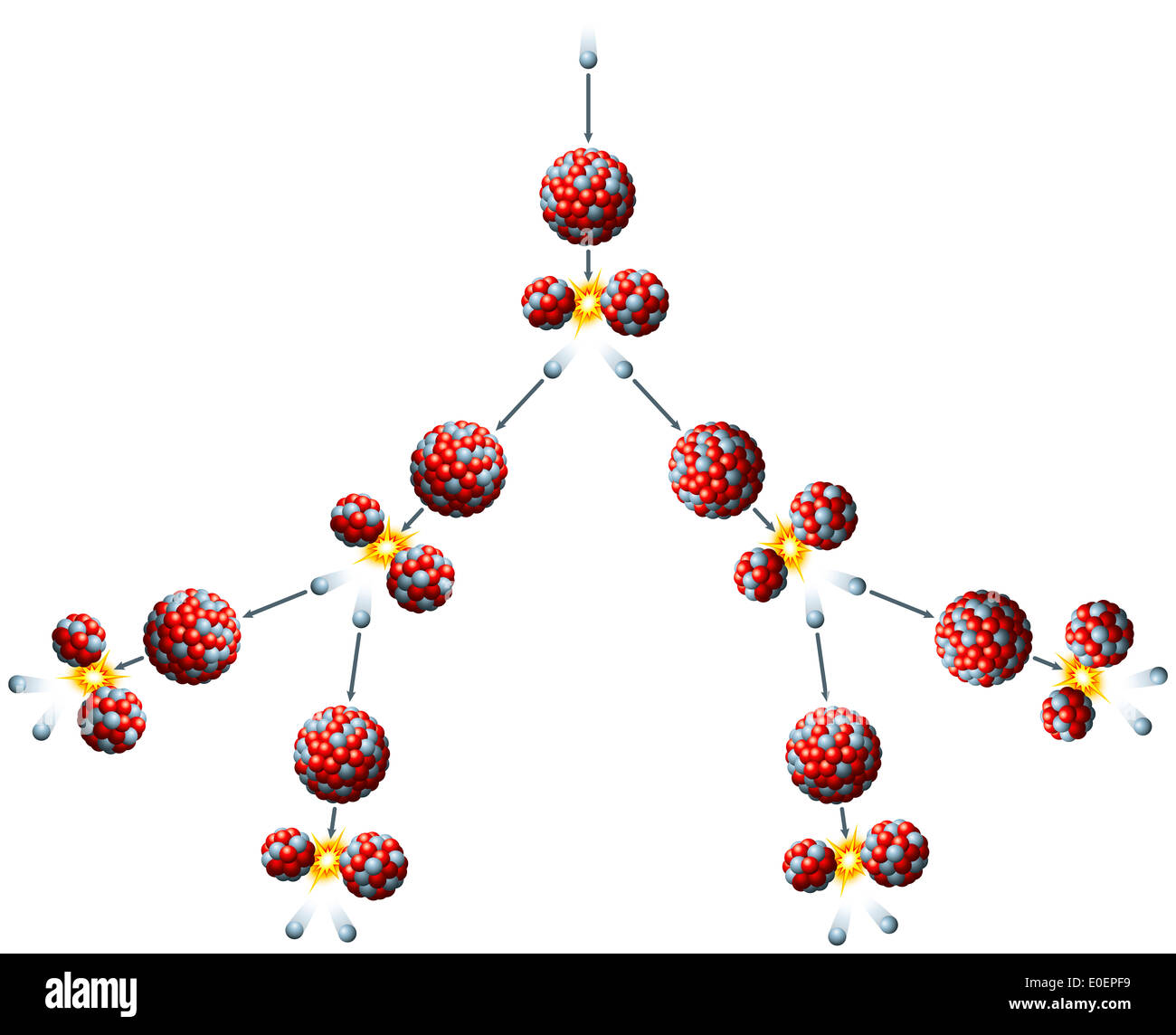
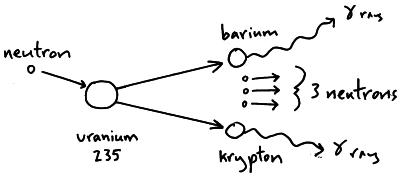
This concentration is insufficient for a self sustaining reaction in a light water reactor enrichment, which just means separating out the uranium-238, must take place to get a usable concentration of uranium-235. Only around 0.72% of all natural uranium is uranium-235, the rest being mostly uranium-238. For fast neutrons it is in the order of 1 barn. The nuclear cross section for slow thermal neutrons is about 1000 barns. However, approximately 5% of this energy is carried away by virtually undetectable neutrinos. The fission of one atom of U-235 generates 200 MeV = 3.2 × 10 -11 J, i.e. In nuclear bombs, the reaction is uncontrolled and the large amount of energy released creates a nuclear explosion. In nuclear reactors, the reaction is slowed down by the addition of control rods which are made of elements such as boron, cadmium, and hafnium which can absorb a large number of neutrons. Some of them produce neutrons, called delayed neutrons, which contribute to the fission chain reaction. A fission chain reaction produces intermediate mass fragments which are highly radioactive and produce further energy by their radioactive decay. A critical chain reaction can be achieved at low concentrations of U-235 if the neutrons from fission are moderated to lower their speed, since the probability for fission with slow neutrons is greater. If the reaction will sustain itself, it is said to be critical, and the mass of U-235 required to produce the critical condition is said to be a critical mass. If at least one neutron from U-235 fission strikes another nucleus and causes it to fission, then the chain reaction will continue. Better weighing performance in 6 easy steps
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
